Innately Trained
On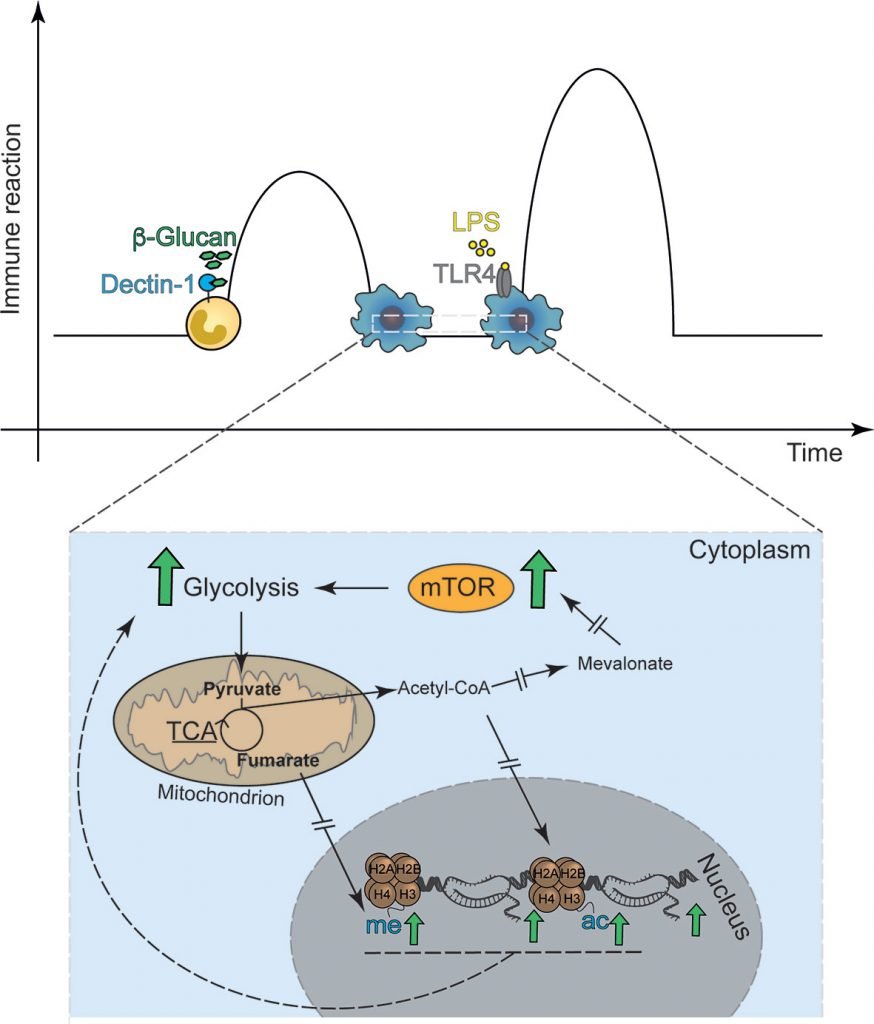
The above illustration1 represents one of the most important recent developments in immunology. It has radically changed the way we think about the innate immune system, and immunity in general.
It turns out that the innate immune system has a memory2. This is not like the memory of the adaptive immune system, which can be life-long; the bulk of innate immune memory lasts for only a few days, although certain aspects of it may remain for a few years3. But it is critically important, and it can be trained.
Positive innate immune training enhances health, and dietary tools can be used to achieve this. Negative training leads to disease, and dietary components play a role here too.
For non-scientists, a very basic intro.
The innate immune system is the body’s first line of defence against potential pathogens. It consists of physical barriers, antimicrobial peptides, antimicrobial enzymes including lactoperoxidase (subject of a future post), and innate immune cells. These include neutrophils, natural killer cells, mast cells, macrophages and a half dozen others, each of which contain sub-populations with different but coordinating roles.
The adaptive immune system is generally not activated unless pathogens manage to bypass the first line of defence and enter the blood and deeper tissues in significant numbers. At this point adaptive immune cells, informed by the innate cells, start to mount a slower but more specific response. They have the ability to form a longer-term memory of that pathogen so that it can be more effectively countered at a subsequent meeting, decades later. This is the basis of immunisation.
Partly because immunisation has become such an important source of revenue to Big Pharma, and partly because it was believed that the innate immune system was a constant, innate immunity has been rather ignored. Until very recently.
In the last 15 years evidence has emerged that Pathogen-Associated Molecular Patterns (PAMPs), found on the surface of different types of pathogens, induce epigenetic changes in innate immune cells4-6 which up-regulate their ability to respond to future pathogens. They do this partly by increasing innate immune cells’ ability to generate energy, via up-regulated glycolysis7-9.
The cells are then able to migrate more quickly (chemotaxis) to the site of an infection, and behave more aggressively towards the pathogens (phagocytosis) when they arise at the site of infection. Accelerated chemotaxis and enhanced phagocytosis mean a more effective acute inflammatory response, and improved resistance to many different pathogens. There are knock-on improvements in adaptive immune function alsoie 10.
This innate enhancement is thought to last as long as the epigenetically re-engineered immune cells last. Macrophages live up to several years11 but monocytes live only for a day or so11 and neutrophils, the most numerous innate immune cells, have a half-life in the blood of 6-8 hours12. It is probably best therefore to prime the innate immune system on a daily basis. In an earlier and less sanitised age, this is exactly what happened; see below.
The 1-3, 1-6 beta glucans are arguably among the most important PAMPs.
Their importance is revealed by the fact that they are recognised by at least 6 different classes of receptors13 present on all innate immune cells (and some T-cells), and enhance key aspects of innate immune function in every animal species tested so far14.
The fact that 1-3, 1-6 beta glucan supplements improve resistance to infection15-18, reduce allergy19-21 and exert significant anti-cancer effects22 indicates that we are not consuming optimal amounts of them. If we were, beta glucan supplements would have no effect. And given that diets low in beta glucans are negatively impacting our personal and public health, it seems fair to classify 1-3, 1-6 beta glucans as essential if atypical nutrients.
Lipopolysaccharides, which are structural elements in the cell walls of gram-negative bacteria, are another well-characterised PAMP. They trigger intense inflammation which, unlike the immune training induced by the beta glucans, becomes destructive if exposure is high and sustained. This occurs in endotoxaemia, and is one of the mechanisms whereby a low-fibre (ie Western) diet increases the risk of many degenerative diseases.
It is not just PAMPs that train the innate immune system. There are also DAMPs, which exert similarly positive effects; and LAMPs, which appear to be entirely negative.
Damaged tissue produces specific Danger-or Damage-Associated Molecular Patterns (DAMPs), some of which are termed ’alarmins’. These behave rather like PAMPs and generally lead, as PAMPs do, to a more effective subsequent immune response22, and to various sickness behaviours23. This is quite logical, as tissue damage would generally allow the ingress of pathogens into deeper tissues; and the perception of danger might well portend the same thing.
Don’t confuse PAMPs and DAMPs with AMPs, anti-microbial proteins which kill pathogens and are essential components in the innate immune system. These are produced by our immune cells when supplied with vitamin D25, and by probiotic species in our intestines when these are fed with prebiotic fiber26, 75.
iDAMPs and cDAMPs, which relate to cell stress and death pathways, are fascinating but outside the scope of this post.
In marked contrast the LAMPs, Lifestyle-Associated Molecular Patterns28, induce innate mal-training. These include such familiar toxins as oxidised LDL cholesterol and monosodium urate (ie gout crystals). Levels of the former are increased by a diet low in antioxidants such as the amphiphile polyphenols29, levels of the latter are increased by a high purine intake; raised levels of both are linked to an increased risk of disease and early death30, 31.
Other candidate LAMPs include proteins damaged by carbamylation, proteins damaged by glycation (AGEs) and advanced lipoxidation end-products (ALEs).
Immune cells are not very effective at clearing LAMPSie 28, so if you persist with an unhealthy lifestyle, they accumulate in the body31. This triggers the inflammasome, causing persistent leucocyte hyperactivation and (sterile) chronic inflammation, which drives progressive damage to the extra-cellular matrix1, 28, 32, 33. This, as blog readers may recall, contributes to ageing and to illness34.
Carbamylated proteins are increased by smoking, and by under-hydration35. AGEs and ALEs, which are produced when foods are cooked at high temperatures, occur at high levels in many ultra-processed foods36-38.
The negative effects of LAMPs seem to be quite long-lasting. One pre-clinical study stands out. In atherosclerosis-prone Ldlr-/- mice, 4 weeks of a Western-style diet epigenetically re-programmed various innate immune cells so that the mice developed exaggerated inflammatory responses, which persisted even after a further 4 weeks of healthy eating39.
To recap: exposure to LAMPS is increased by ultra-processed foods and by the Western-type diet in general, which is rich in fats and sugars and low in prebiotic fibres and the 1-3, 1-6 beta glucans. Such a diet is also depleted in antioxidants and anti-inflammatory nutrients. This toxic combination inevitably leads to chronic inflammation, progressive tissue damage, downgraded immunity and increased rates of chronic degenerative diseases such as cancer.
This situation is made worse by our substantially reduced dietary intakes of the key dietary PAMP, the 1-3, 1-6 beta glucans.
The idea that the removal of these beta glucans has made us more vulnerable to cancer derives from studies documenting these compounds’ distinct cancer-fighting properties, many of which are related to innate immune training.
In ‘successful’ cancers, host immune cells are recruited by the tumor cells to alter the immune cell phenotype/function and tricked into immune suppression or tolerance40, 41. The beta glucans are thought to be able to switch the suppressed immune cells back on via innate training, thus restoring local cancer-hostile immunity42-44.
This is why the beta glucans’ ability to induce positively trained innate immunity is now being suggested as a potentially important anti-cancer strategy42; and why there are so many recent and on-going pre-clinical and clinical cancer trials43.
Unfortunately, the 1-3, 1-6 beta glucans have largely disappeared from our diet.
These structural carbohydrate compounds occur only in the cell walls of fungi, and the most common fungal food sources are yeast and mushrooms. Due to the different cell wall requirements of unicellular yeast and multicellular mushrooms, beta glucans from these two sources have different forms. These include lengths of the 1-3 side chains, 1-6 chain lengths and tertiary helical and annular structures44-48.
Yeast beta glucans are more potent innate immune primers in some assays and mushroom-derived glucans do better in others, but I generally favour yeast derivates. My simplistic thinking is that our innate immune systems were designed to fight off yeasts and other single cell fungi, rather than truffles.
Yeasts, some of the most pervasive members of the fungal kingdomie 49, were formally omnipresent in our diet. They were present in large amounts in fermented foods such as beer, wine and breads, and at lower levels as a background contaminant in almost all other foods50.
Prior to the 1950’s the natural fungicides used in agriculture were not particularly effective, so that fungal residues were present in practically everything we ate. The more potent synthetic compounds which started to became common usage in the 50’s reduced fungal spoilage considerably, reducing yeast contamination on fruits, grains and vegetables51.
Food production chains have been tightened to minimize contamination and spoilage, and improve quality. Today crops are collected and, in some cases, flash-frozen within minutes of harvesting52. The effective abolition of storage time reduces background levels of yeast even further.
Processing changes have also impacted the yeast-fermented beverages. Filtration of beer was introduced by the German engineer Lorenz Enzinger at a brewing trade show in 1880, designed to produce beer that was abiotic and had better storage qualities. The technology graduated from paper and cotton filters to kieselguhr and finally, in the 1960’s, to ultra-filtration using cross-flow membranes.
These produce a sterile beer that keeps for months but contains only homeopathic amounts of beta glucan. The same filtration process removes the sediments from wines (decanters have become an affectation) and so the lees, and the beta glucans, have been removed from these too.
Breads and other baked goods still contain beta glucans but we eat far less of these foods than did our ancestors, due to our reduced calorific requirements53.
While it is hard to quantify the effects on our health of the reduction in our intake of 1-3, 1-6 beta glucans, there is some evidence that we may have become more vulnerable to developing sepsis54, although this apparent increase may also reflect better diagnosis55. The fact that cancers are emerging in progressively younger groups of patients56 could be another sign of impaired immunosurveillance42-44.
It certainly looks as if paediatric health is impacted. Yest-derived beta glucans are routinely added to infant formula, because they have been shown to reduce rotavirus infection57. In other words, beta glucan-depleted toddlers are immunologically weaker and more prone to infection.
It is time to put these valuable natural compounds back in all our foods, where they used to be. It makes sense also to use them to train the innate immune systems of elective surgery patients, because the hospital environments where they will be operated on are hotbeds of antibiotic-resistant bacteria58, 59.
Beta glucans are not the only PAMPs but they are safe, inexpensive and widely available.
And, they have a feel-good effect. Among the many innate immune cells they train are the microglia, the primary innate immune cells in the brain60. The resulting changes in microglial function which include physiological (ie low level) neuroinflammation60-62 may explain the improvement in mood that has been consistently linked to beta glucan use63-66.
Equally important, there is emerging evidence that the ability of the beta glucans to induce physiological levels of neuroinflammation facilitates neuronal repair67, 68, enhances various aspects of intelligence69, and likely reduces the risk of neurodegenerative disease70.
Taking a 1-3, 1-6 beta glucan supplement seems like an innately smart thing to do. While you’re at it, take a 1-3, 1-4 beta glucan supplement to transform your dysbiotic gut into another functional part of your innate immune system.
Next week: Middle C, and the music of meat.
References
- Jentho E, Weis S. DAMPs and Innate Immune Training. Front Immunol. 2021 Oct 22;12:699563.
- Netea MG, Quintin J, van der Meer JW. Trained immunity: a memory for innate host defense. Cell Host Microbe. 2011 May 19;9(5):355-61.
- Netea MG, Dominguez-Andres J, Barreiro LB, Chavakis T, Divangahi M, Fuchs E, Joosten LAB, van der Meer JWM, Mhlanga MM, Mulder WJM, et al. Defining trained immunity and its role in health and disease. Nat Rev Immunol. 2020;20(6):375–88.
- Saeed S, Quintin J, Kerstens HH, Rao NA, Aghajanirefah A, Matarese F, Cheng SC, Ratter J, Berentsen K, van der Ent MA, Sharifi N, Janssen-Megens EM, Ter Huurne M, Mandoli A, van Schaik T, Ng A, Burden F, Downes K, Frontini M, Kumar V, Giamarellos-Bourboulis EJ, Ouwehand WH, van der Meer JW, Joosten LA, Wijmenga C, Martens JH, Xavier RJ, Logie C, Netea MG, Stunnenberg HG. Epigenetic programming of monocyte-to-macrophage differentiation and trained innate immunity. Science. 2014 Sep 26;345(6204):1251086.
- Quintin J, Saeed S, Martens JHA, Giamarellos-Bourboulis EJ, Ifrim DC, Logie C, Jacobs L, Jansen T, Kullberg BJ, Wijmenga C, Joosten LAB, Xavier RJ, van der Meer JWM, Stunnenberg HG, Netea MG. Candida albicans infection affords protection against reinfection via functional reprogramming of monocytes. Cell Host Microbe. 2012 Aug 16;12(2):223-32.
- Kleinnijenhuis J, Quintin J, Preijers F, Joosten LA, Jacobs C, Xavier RJ, van der Meer JW, van Crevel R, Netea MG. BCG-induced trained immunity in NK cells: Role for non-specific protection to infection. Clin Immunol. 2014 Dec;155(2):213-9.
- Domínguez-Andrés J, Novakovic B, Li Y, Scicluna BP, Gresnigt MS, Arts RJW, Oosting M, Moorlag SJCFM, Groh LA, Zwaag J, Koch RM, Ter Horst R, Joosten LAB, Wijmenga C, Michelucci A, van der Poll T, Kox M, Pickkers P, Kumar V, Stunnenberg H, Netea MG. The Itaconate Pathway Is a Central Regulatory Node Linking Innate Immune Tolerance and Trained Immunity. Cell Metab. 2019 Jan 8;29(1):211-220.e5.
- Arts RJ, Novakovic B, Ter Horst R, Carvalho A, Bekkering S, Lachmandas E, Rodrigues F, Silvestre R, Cheng SC, Wang SY, Habibi E, Gonçalves LG, Mesquita I, Cunha C, van Laarhoven A, van de Veerdonk FL, Williams DL, van der Meer JW, Logie C, O’Neill LA, Dinarello CA, Riksen NP, van Crevel R, Clish C, Notebaart RA, Joosten LA, Stunnenberg HG, Xavier RJ, Netea MG. Glutaminolysis and Fumarate Accumulation Integrate Immunometabolic and Epigenetic Programs in Trained Immunity. Cell Metab. 2016 Dec 13;24(6):807-819.
- Bekkering S, Arts RJW, Novakovic B, Kourtzelis I, van der Heijden CDCC, Li Y, Popa CD, Ter Horst R, van Tuijl J, Netea-Maier RT, van de Veerdonk FL, Chavakis T, Joosten LAB, van der Meer JWM, Stunnenberg H, Riksen NP, Netea MG. Metabolic Induction of Trained Immunity through the Mevalonate Pathway. Cell. 2018 Jan 11;172(1-2):135-146.e9.
- Kirmaz C, Bayrak P, Yilmaz O, Yuksel H. Effects of glucan treatment on the Th1/Th2 balance in patients with allergic rhinitis: a double-blind placebo-controlled study. Eur Cytokine Netw. 2005 Jun;16(2):128-34.
- van Furth R, Cohm ZA. The origin and kinetics of mononuclear phagocytes. J Exp Med. 1968;128:415–435.
- Summers C, Rankin SM, Condliffe AM, Singh N, Peters AM, Chilvers ER. Neutrophil kinetics in health and disease. Trends Immunol. 2010 Aug;31(8):318-24.
- Motta F, Gershwin ME, Selmi C. Mushrooms and immunity. J Autoimmun. 2021 Feb;117:102576.
- Vetvicka V, Teplyakova TV, Shintyapina AB, Korolenko TA. Effects of Medicinal Fungi-Derived β-Glucan on Tumor Progression. J Fungi (Basel). 2021 Mar 25;7(4):250.
- Dellinger EP, Babineau TJ, Bleicher P, Kaiser AB, Seibert GB, Postier RG, Vogel SB, Norman J, Kaufman D, Galandiuk S, Condon RE. Effect of PGG-glucan on the rate of serious postoperative infection or death observed after high-risk gastrointestinal operations. Betafectin Gastrointestinal Study Group. Arch Surg. 1999 Sep;134(9):977-83.
- Babineau TJ, Marcello P, Swails W, Kenler A, Bistrian B, Forse RA. Randomized phase I/II trial of a macrophage-specific immunomodulator (PGG-glucan) in high-risk surgical patients. Ann Surg. 1994 Nov;220(5):601-9.
- Tzianabos AO, Cisneros RL. Prophylaxis with the immunomodulator PGG glucan enhances antibiotic efficacy in rats infected with antibiotic-resistant bacteria. Ann N Y Acad Sci. 1996 Oct 25;797:285-7.
- Samuelsen AB, Schrezenmeir J, Knutsen SH. Effects of orally administered yeast-derived beta-glucans: a review. Mol Nutr Food Res. 2014 Jan;58(1):183-93.
- Mahmood F, Hetland G, Nentwich I, Mirlashari MR, Ghiasvand R, Nissen-Meyer LSH. Agaricus blazei-Based Mushroom Extract Supplementation to Birch Allergic Blood Donors: A Randomized Clinical Trial. Nutrients. 2019 Oct 2;11(10):2339.
- Jesenak M, Hrubisko M, Majtan J, Rennerova Z, Banovcin P. Anti-allergic effect of Pleuran (β-glucan from Pleurotus ostreatus) in children with recurrent respiratory tract infections. Phytother Res. 2014 Mar;28(3):471-4.
- Sarinho E, Medeiros D, Schor D, Rego Silva A, Sales V, Motta ME, Costa A, Azoubel A, Rizzo JA. Production of interleukin-10 in asthmatic children after Beta-1-3-glucan. Allergol Immunopathol (Madr). 2009 Jul-Aug;37(4):188-92.
- Chen GY, Nuñez G. Sterile inflammation: sensing and reacting to damage. Nat Rev Immunol. 2010 Dec;10(12):826-37.
- https://whitesmoke-heron-286383.hostingersite.com/blog/moral-fiber/
- Geller A, Shrestha R, Yan J. Yeast-Derived β-Glucan in Cancer: Novel Uses of a Traditional Therapeutic. Int J Mol Sci. 2019 Jul 24;20(15):3618.
- Subramanian K, Bergman P, Henriques-Normark B. Vitamin D Promotes Pneumococcal Killing and Modulates Inflammatory Responses in Primary Human Neutrophils. J Innate Immun. 2017;9(4):375-386.
- Negi YK, Pandey C, Saxena N, Sharma S, Garg FC, Garg SK. Isolation of antibacterial protein from Lactobacillus spp. and preparation of probiotic curd. J Food Sci Technol. 2018 Jun;55(6):2011-2020.
- Amiri S, Mokarram RR, Khiabani MS, Bari MR, Khaledabad MA. Characterization of antimicrobial peptides produced by Lactobacillus acidophilus LA-5 and Bifidobacterium lactis BB-12 and their inhibitory effect against foodborne pathogens. LWT Volume 153, January 2022, 112449
- Zindel J, Kubes P. DAMPs, PAMPs, and LAMPs in Immunity and Sterile Inflammation. Annu Rev Pathol. 2020 Jan 24;15:493-518.
- EFSA Journal 2011;9(4):2033. https://efsa.onlinelibrary.wiley.com/doi/pdf/10.2903/j.efsa.2011.2033 Accessed 15.7.23
- Fisher MC, Rai SK, Lu N, Zhang Y, Choi HK. The unclosing premature mortality gap in gout: a general population-based study. Ann Rheum Dis. 2017 Jul;76(7):1289-1294.
- Chen C, Lü JM, Yao Q. (2016). Hyperuricemia-related diseases and xanthine oxidoreductase (XOR) inhibitors: an overview. Med. Sci. Monit. 22, 2501–2512.
- Semba RD, Nicklett EJ, Ferrucci L. Does accumulation of advanced glycation end products contribute to the aging phenotype? J Gerontol A Biol Sci Med Sci. 2010 Sep;65(9):963-75.
- Jové M, Mota-Martorell N, Pradas I, Martín-Gari M, Ayala V, Pamplona R. The Advanced Lipoxidation End-Product Malondialdehyde-Lysine in Aging and Longevity. Antioxidants (Basel). 2020 Nov 15;9(11):1132.
- https://whitesmoke-heron-286383.hostingersite.com/blog/matrix-reloaded/
- https://whitesmoke-heron-286383.hostingersite.com/blog/dry-spell/
- Goldberg T, Cai W, Peppa M, Dardaine V, Baliga BS, Uribarri J, Vlassara H. Advanced glycoxidation end products in commonly consumed foods. J Am Diet Assoc. 2004 Aug;104(8):1287-91.
- Uribarri J, Woodruff S, Goodman S, Cai W, Chen X, Pyzik R, Yong A, Striker GE, Vlassara H. Advanced glycation end products in foods and a practical guide to their reduction in the diet. J Am Diet Assoc. 2010 Jun;110(6):911-16
- Gil A, Bengmark S. Productos finales de la glicación y de la lipoxidación como amplificadores de la inflamación: papel de los alimentos [Advanced glycation and lipoxidation end products–amplifiers of inflammation: the role of food]. Nutr Hosp. 2007 Nov-Dec;22(6):625-40. Spanish.
- Christ A, Günther P, Lauterbach MAR, Duewell P, Biswas D, Pelka K, Scholz CJ, Oosting M, Haendler K, Baßler K, Klee K, Schulte-Schrepping J, Ulas T, Moorlag SJCFM, Kumar V, Park MH, Joosten LAB, Groh LA, Riksen NP, Espevik T, Schlitzer A, Li Y, Fitzgerald ML, Netea MG, Schultze JL, Latz E. Western Diet Triggers NLRP3-Dependent Innate Immune Reprogramming. Cell. 2018 Jan 11;172(1-2):162-175.e14.
- Whiteside TL. The tumor microenvironment and its role in promoting tumor growth. Oncogene. 2008 Oct 6;27(45):5904-12.
- Croci DO, Zacarías Fluck MF, Rico MJ, Matar P, Rabinovich GA, Scharovsky OG. Dynamic cross-talk between tumor and immune cells in orchestrating the immunosuppressive network at the tumor microenvironment. Cancer Immunol Immunother. 2007 Nov;56(11):1687-700.
- Geller A, Shrestha R, Yan J. Yeast-Derived β-Glucan in Cancer: Novel Uses of a Traditional Therapeutic. Int J Mol Sci. 2019 Jul 24;20(15):3618.
- Zhang M, Kim JA, Huang AY. Optimizing Tumor Microenvironment for Cancer Immunotherapy: β-Glucan-Based Nanoparticles. Front Immunol. 2018 Feb 26;9:341.
- Li D, Li W, Zheng P, Yang Y, Liu Q, Hu Y, He J, Long Q, Ma Y. A “trained immunity” inducer-adjuvanted nanovaccine reverses the growth of established tumors in mice. J Nanobiotechnology. 2023 Mar 2;21(1):74.
- Bohn JA, BeMiller JN. (1→3)-β-d-Glucans as biological response modifiers: A review of structure-functional activity relationships. Carbohydr. Polym. 1995;28:3–14.
- Šandula J., Kogan G., Kačuráková M., Machová E. Microbial (1→3)-β-d-glucans, their preparation, physico-chemical characterization and immunomodulatory activity. Carbohydr. Polym. 1999;38:247–253.
- Meng X, Liang H, Luo L. Antitumor polysaccharides from mushrooms: a review on the structural characteristics, antitumor mechanisms and immunomodulating activities. Carbohydr Res. (2016) 424:30–41.
- Manabe N, Yamaguchi Y. 3D Structural Insights into β-Glucans and Their Binding Proteins. Int J Mol Sci. 2021 Feb 4;22(4):1578.
- Snyder AB, Worobo RW. Fungal Spoilage in Food Processing. J Food Prot. 2018 Jun;81(6):1035-1040.
- Hernández A, Pérez-Nevado F, Ruiz-Moyano S, Serradilla MJ, Villalobos MC, Martín A, Córdoba MG. Spoilage yeasts: What are the sources of contamination of foods and beverages? Int J Food Microbiol. 2018 Dec 2;286:98-110.
- Morton V, Staub T. 2008 A Short History of Fungicides. https://www.apsnet.org/edcenter/apsnetfeatures/Pages/Fungicides.aspx Accessded 5.6.23
- https://www.paradise-fruits.de/en/products/iqf-fruits/ Accessed 8.6.23
- Clayton P, Rowbotham J. How the mid-Victorians worked, ate and died. Int J Environ Res Public Health. 2009 Mar;6(3):1235-53.
- Kramarow EA. Sepsis-related Mortality Among Adults Aged 65 and Over: United States, 2019. NCHS Data Brief. 2021 Nov;(422):1-8.
- Rhee C, Klompas M. Sepsis trends: increasing incidence and decreasing mortality, or changing denominator? J Thorac Dis. 2020 Feb;12(Suppl 1):S89-S100.
- Ugai T, Sasamoto N, Lee HY, Ando M, Song M, Tamimi RM, Kawachi I, Campbell PT, Giovannucci EL, Weiderpass E, Rebbeck TR, Ogino S. Is early-onset cancer an emerging global epidemic? Current evidence and future implications. Nat Rev Clin Oncol. 2022 Oct;19(10):656-673.
- Li F, Jin X, Liu B, Zhuang W, Scalabrin D. Follow-up formula consumption in 3- to 4-year-olds and respiratory infections: an RCT. Pediatrics. 2014 Jun;133(6):e1533-40.
- Cruz R, Romalde JL, Nesme J, Sørensen SJ, Smets BF, Graham D, Paul M. Comparison of antibiotic-resistant bacteria and antibiotic resistance genes abundance in hospital and community wastewater: A systematic review. Sci Total Environ. 2020 Nov 15;743:140804.
- Antibiotic Resistant Germs in Hospitals: Information for Patients and their Families. CDC 2019 https://www.cdc.gov/hai/patientsafety/ar-hospitals.html Accessed 16.7.23
- Heng Y, Zhang X, Borggrewe M, van Weering HRJ, Brummer ML, Nijboer TW, Joosten LAB, Netea MG, Boddeke EWGM, Laman JD, Eggen BJL. Systemic administration of β-glucan induces immune training in microglia. J Neuroinflammation. 2021 Feb 22;18(1):57.
- Shah VB, Huang Y, Keshwara R, Ozment-Skelton T, Williams DL, Keshvara L. Beta-glucan activates microglia without inducing cytokine production in Dectin-1-dependent manner. J Immunol. 2008 Mar 1;180(5):2777-85.
- Shah VB, Williams DL, Keshvara L. beta-Glucan attenuates TLR2- and TLR4-mediated cytokine production by microglia. Neurosci Lett. 2009 Jul 24;458(3):111-5.
- Talbott SM, Talbott JA. Baker’s yeast beta-glucan supplement reduces upper respiratory symptoms and improves mood state in stressed women. J Am Coll Nutr. 2012 Aug;31(4):295-300.
- Talbott S, Talbott J. Effect of BETA 1, 3/1, 6 GLUCAN on Upper Respiratory Tract Infection Symptoms and Mood State in Marathon Athletes. J Sports Sci Med. 2009 Dec 1;8(4):509-15.
- Talbott S. Beta 1,3/1,6 glucan decreases upper respiratory tract infection symptoms and improves psychological well-being in moderate to highly-stressed subjects. Jan 2010 Agro Food Industry Hi Tech 21(1):21-24
- Vlassopoulou M, Yannakoulia M, Pletsa V, Zervakis GI, Kyriacou A. Effects of fungal beta-glucans on health – a systematic review of randomized controlled trials. Food Funct. 2021 Apr 26;12(8):3366-3380.
- Baldwin KT, Carbajal KS, Segal BM, Giger RJ. Neuroinflammation triggered by β-glucan/dectin-1 signaling enables CNS axon regeneration. Proc Natl Acad Sci U S A. 2015 Feb 24;112(8):2581-6.
- Zhang D, Jia Y, Sun X, Li H, Yin M, Li H, Dai L, Han L, Wang L, Qian M, Du J, Zhu K, Bao H. The Dectin-1 Receptor Signaling Pathway Mediates the Remyelination Effect of Lentinan through Suppression of Neuroinflammation and Conversion of Microglia. J Immunol Res. 2022 Dec 28;2022:3002304.
- Hu M, Zhang P, Wang R, Zhou M, Pang N, Cui X, Ge X, Liu X, Huang XF, Yu Y. Three Different Types of β-Glucans Enhance Cognition: The Role of the Gut-Brain Axis. Front Nutr. 2022 Mar 3;9:848930.
- https://whitesmoke-heron-286383.hostingersite.com/blog/diet-drugs-and-dementia/


